Overview
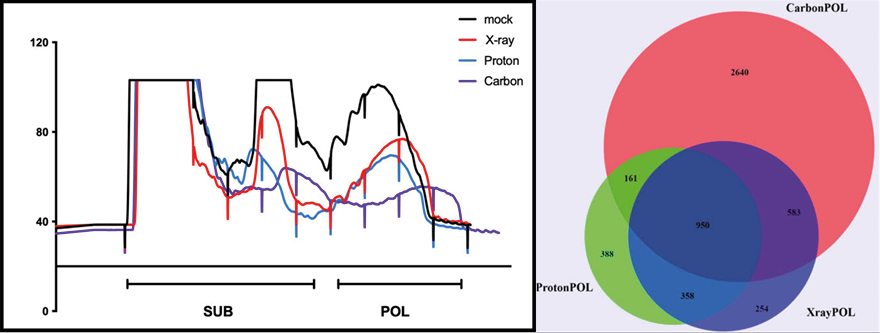
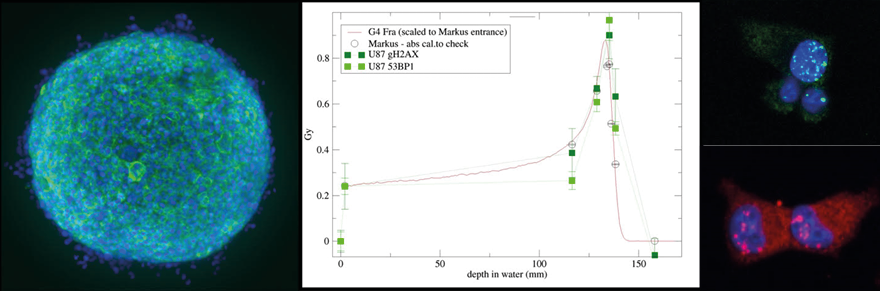
Radiation therapy is an important tool for cancer treatment and approximately 50% of all malignant cancer patients receive radiation therapy during their course of illness. The aim of radiotherapy is to destroy efficiently cancer cells with ionizing radiation causing severe DNA damage resulting in cancer cell death while minimizing exposure to nearby healthy tissues and organs at risk. The most common radiotherapy employs X-rays, but also charged particle accelerated by a synchrotron or cyclotron can be used for patient treatment. With the introduction of charged particle radiotherapy (proton therapy), the area of irradiated healthy tissue surrounding the tumor was further decreased.
The main focus of the laboratory is the characterization of the different biological mechanisms activated by radiotherapy, in particular Proton therapy, in cancer and normal cells.
We are using different cellular models such as 2D cell culture, 3D spheroids, and Scaffold-based cultures in order to better understand tumor and healthy tissue responses to radiation therapy.
Research Projects
- p21 as a main player in cancer cells radioresistance.
- Unraveling the molecular mechanisms underlying the abscopal effect.
- Development and characterization of innovative 3D in vitro models.
- Biological characterization of FLASH radiotherapy using high dose-rate particle beam.
Group members
- Alessandra Bisio, PI
- Laura Bartolomei, PhD Student
- Samuele Bugnara, Master Student
Collaborations
- Emanuele Scifoni, TIFPA - Trento Institute for Fundamental Physics and Applications, Trento, Italy
- Juergen Borlak, Hannover Medical School (MHH), Germany
- Alberto Inga, Department of Cellular, Computational and Integrative Biology - CIBIO, University of Trento, Trento, Italy
- Walter Tinganelli, GSI Helmholtz Centre for Heavy Ion Research, Darmstadt, Germany
- Devid Maniglio, Department of Industrial Engineering, University of Trento, Italy
- Takashi Shimohkawa, NIRS- National Institute of Radiological Sciences, Chiba, Japan
- Antonella Motta, Department of Industrial Engineering, University of Trento, Italy
- Ira-Ida Skvortsova Medical University of Innsbruck Tyrolean Cancer Research Institute, Innsbruck, Austria
- Palmina Simoniello, Università Parthenope, Napoli, Italy
- Francesco Cammarata, Giorgio Russo, CNR-IBFM, Cefalù, Italy
Selected publications
- Bosco B, Rossi A, Rizzotto D, Hamadou M, Bisio A, Giorgetta S, Perzolli A, Bonollo F, Gaucherot A, Catez F, Diaz JJ, Dessi E, Inga A. DHX30Coordinates Cytoplasmic Translation and Mitochondrial Function Contributing to Cancer Cell Sur-vival. Cancers, MDPI, 2021, 13, 10.3390/cancers13174412
- Galeaz C, Totis C, Bisio A. Radiation Resistance: A Matter of Transcription Factors. Front Oncol. 2021 Jun 1;11:662840. doi: 10.3389/fonc.2021.662840. eCollection 2021.
- Alaimo A, Lorenzoni M, Ambrosino P, Bertossi A, Bisio A, Macchia A, Zoni E, Genovesi S, Cambuli F, Foletto V, De Felice D, Soldovieri MV, Mosca I, Gandolfi F, Brunelli M, Petris G, Cereseto A, Villarroel A, Thalmann G, Carbone FG, Kruithof-de Julio M, Barbareschi M, Romanel A, Taglialatela M, Lunardi A. Calcium cytotoxicity sensitizes prostate cancer cells to standard-of-care treatments for locally advanced tumors. Cell Death Dis. 2020 Dec 7;11(12):1039. doi: 10.1038/s41419-020-03256-5.
- Ballan M, Tosato M, Verona M, Caeran M, Borgna F, Vettorato E, Corradetti S, Zangrando L, Sgaravatto M, Verlato M, Asti M, Marzaro G, Mastrotto F, Di Marco V, Maniglio D, Bisio A, Motta A, Quaranta A, Zenoni A, Pastore P, Realdon N, Andrighetto A. Preliminary evaluation of the production of non-carrier added 111Ag as core of a therapeutic radiopharmaceutical in the framework of ISOLPHARM_Ag experiment. Appl Radiat Isot. 2020 Oct; 164:109258. doi: 10.1016/j.apradiso.2020.109258.
- Ebner DK, Tinganelli W, Helm A, Bisio A, Simoniello P, Natale F, et al. Generating and grading the abscopal effect: Proposal for comprehensive evaluation of combination immunoradiotherapy in mouse models. Translational Cancer Research. 2017;6:S892-S9. doi: 10.21037/tcr.2017.06.01
- Ebner DK, Tinganelli W, Helm A, Bisio A, Yamada S, Kamada T, et al. The immunoregulatory potential of particle radiation in cancer therapy. Frontiers in Immunology. 2017; Feb 6;8:99. doi: 10.3389/fimmu.2017.00099. eCollection 2017.
- Andreotti V*, Bisio A*, Bressac-de Paillerets B, Harland M, Cabaret O, Newton-Bishop J, et al. The CDKN2A/p16INK4a5'UTR sequence and translational regulation: Impact of novel variants predisposing to melanoma. Pigment Cell and Melanoma Research. 2016;29(2):210-21. doi: 10.1111/pcmr.12444
- Sharma V, Jordan JJ, Ciribilli Y, Resnick MA, Bisio A§, Inga A§. Quantitative analysis of NF-κB transactivation specificity using a yeast-based functional assay. PLoS ONE. 2015;10(7). doi: 10.1371/journal.pone.0130170
- Bisio A, Latorre E, Andreotti V, Paillerets BB, Harland M, Scarra GB, et al. The 5'-untranslated region of p 16INK4amelanoma tumor suppressor acts as a cellular IRES, controlling mRNA translation under hypoxia through YBX1 binding. Oncotarget. 2015;6(37):39980-94. doi: 10.18632/oncotarget.5387
- Zaccara S, Tebaldi T, Pederiva C, Ciribilli Y, Bisio A, Inga A. P53-directed translational control can shape and expand the universe of p53 target genes. Cell Death and Differentiation. 2014;21(10):1522-34. doi: 10.1038/cdd.2014.79
- Bisio A, Zámborszky J, Zaccara S, Lion M, Tebaldi T, Sharma V, et al. Cooperative interactions between p53 and NFκB enhance cell plasticity. Oncotarget. 2014;5(23):12111-25. doi: 10.18632/oncotarget.2545
- Bisio A, De Sanctis V, Del Vescovo V, Denti MA, Jegga AG, Inga A, et al. Identification of new p53 target microRNAs by bioinformatics and functional analysis. BMC cancer. 2013;13. doi: 10.1186/1471-2407-13-552
- Bisio A, Nasti S, Jordan JJ, Gargiulo S, Pastorino L, Provenzani A, et al. Functional analysis of CDKN2A/p16INK4a5′-UTR variants predisposing to melanoma. Human Molecular Genetics. 2010;19(8):1479-91. doi: 10.1093/hmg/ddq022
