Overview
The laboratory deals with the study of cardiac physiology/pathophysiology through the development of signal analysis techniques, computational modeling, and multimodal imaging. The computational approach is combined with experimental studies in cardiac cellular models. The research is focused on the pathogenesis of cardiac arrhythmias, and it is carried out through collaborations with clinical departments and biomedical industries, with the ultimate goal of defining new biomarkers for cardiac diseases and new therapeutic approaches.
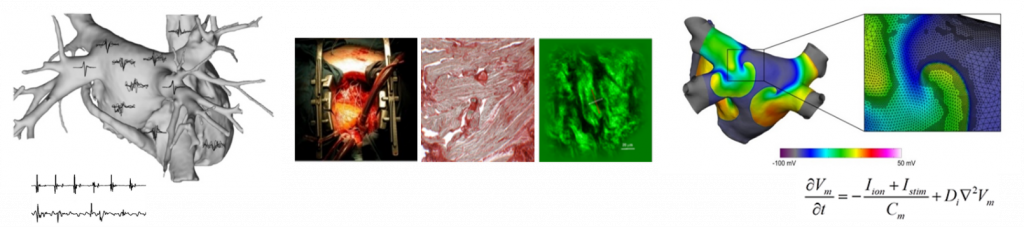
Research directions
- Multiscale investigation of complex arrhythmias
Cardiac arrhythmias are often complex multifactorial diseases where several cardiac and non-cardiac predisposing factors, including genetics, age-/disease-associated remodeling, and autonomic dysfunctions, concur to the formation of arrhythmic triggers and substrates. The incomplete understanding of arrhythmia mechanisms and the multiplicity of factors promoting arrhythmias in humans have hampered the development of effective therapeutic approaches. Our work combines the use of advanced signal analysis, multimodal imaging, and computational models with the structural/molecular analysis of cardiac tissue samples to understand the mechanisms underlying arrhythmia development and to improve therapeutic approaches.
- Proton radiosurgery as new ablation treatment of life-threatening arrhythmias
Ventricular arrhythmias are the leading cause of sudden cardiac death worldwide. Among therapeutic approaches for arrhythmias, proton-based radiosurgery of arrhythmic targets constitutes a development frontier, which holds the promise for minimal invasivity, high precision, and focused energy delivery. In the framework of PNRR-iNEST project and within a research network that includes the Proton Therapy Center, the Cardiology and Radiology Divisions of the APSS of Trento, we are investigating the feasibility of image-guided proton radiosurgery ablation as a new therapeutic modality for patients with life-threatening ventricular tachycardias. Innovative multimodal imaging integration approaches are developed which allow to merge all the information on the structural and electrophysiological substrate of the arrhythmia and to precisely define the target area of the therapy. The extension of the application of radiosurgery with proton beams from the oncological and/or neurosurgical fields to the cardiological one will open new frontiers in the field of radiosurgery of arrhythmias.
- Radiobiological studies in cardiac cellular models
We investigate the radiobiological effects of ionizing radiation on the heart through in vitro studies on cardiac tissue preparations of increasing structural complexity. Changes in the electrical activity of cardiac cells, DNA damage, and cardiomyocyte gene expression are evaluated upon irradiation by proton beams at increasing doses. To characterize the different biological mechanisms activated by proton therapy in cardiac cells and tissues, different in vitro models, such as 2D cell cultures, 3D spheroids, and scaffold-based cultures, are developed. Heart-on-chip high-throughput platforms to test and validate proton therapy will also be realized in collaboration with the BIOtech Center (Univ. of Trento) and companies working in this field. The results of this basic research will be useful for the mechanistic interpretation of the results obtained from the cardiac proton ablation studies in patients.
Group members
- Flavia Ravelli, PI
- Michela Masè, Post-doc
- Alessandro Cristoforetti, Post-doc
- Marianna Ciuffreda, Pre-doc
- Pilar Escribano Cano, visiting PhD student
Collaborations
- BioMIT, Universitat Politècnica de València, Valencia (Spain) (J.J. Rieta)
- Biomedical Engineering Research Group, Dept. of Electronic, University of Castilla-La Mancha (R. Alcaraz Martínez)
- Department of Cardiovascular Sciences, University of Leicester, UK (T. Almeida)
- BIOtech – Center for Biomedical Technologies, University of Trento, Italy (A. Motta, G. Nollo)
- CISMed – University of Trento (C.C. Quattrocchi)
- Divisions of Cardiology, Cardiac Surgery and Radiology, APSS, Trento, Italy
- Proton Therapy Center, APSS, Trento, Italy
- Medicaltech s.r.l., Polo Tecnologico, Rovereto, Italy
Selected publications
Ravelli F, Masè M, Cristoforetti A, Avogaro L, D'Amato E, Tessarolo F, Piccoli F, Graffigna A. Quantitative assessment of transmural fibrosis profile in the human atrium: evidence for a three-dimensional arrhythmic substrate by slice-to-slice histology. Europace 25(2):739-747, 2023
Masè M, Cristoforetti A, Del Greco M and Ravelli F. A divergence-based approach for the identification of atrial fibrillation focal drivers from multipolar mapping: a computational study. Frontiers in Physiology 12, 749430, 2021
Almeida T, Soriano D, Masè M, Ravelli F, Bezerra A, Li X, Chu G, Salinet J, Stafford P, Andre Ng G, Schlindwein F, Yoneyama T. Unsupervised classification of atrial electrograms for electroanatomic mapping of human persistent atrial fibrillation IEEE Transactions on Biomedical Engineering 68(4), art. no. 9186302, pp 1131-1141, 2021
Masè, M, Grasso M, Avogaro L, Giacomaz MN, D'Amato E, Tessarolo F, Graffigna A, Denti MA and Ravelli, F. Upregulation of mir-133b and mir-328 in patients with atrial dilatation: Implications for stretch-induced atrial fibrillation. Frontiers in Physiology 10, art. no. 1133, 2019
Cristoforetti A, Mase M, Bonmassari R, Dallago M, Nollo G and Ravelli F. A patient-specific mass-spring model for biomechanical simulation of aortic root tissue during transcatheter aortic valve implantation. Physics in Medicine and Biology 64 (8), art. no. 085014, 2019
Disertori M, Masè M and Ravelli F. Myocardial fibrosis predicts ventricular tachyarrhythmias. Trends in Cardiovascular Medicine 27 (5), pp. 363-372, 2017 (Review)
Masè M, Grasso M, Avogaro L, D'Amato E, Tessarolo F, Graffigna A, Denti MA and Ravelli F. Selection of reference genes is critical for miRNA expression analysis in human cardiac tissue. A focus on atrial fibrillation. Scientific Reports 7, art. no. 41127, 2017
Disertori M, Rigoni M, Pace N, Casolo G, Masè M, Gonzini L, Lucci, D., Nollo, G. and Ravelli, F. Myocardial fibrosis assessment by LGE is a powerful predictor of ventricular tachyarrhythmias in ischemic and nonischemic LV dysfunction: a meta-analysis. JACC: Cardiovascular Imaging 9 (9), pp. 1046-1055, 2016
Ravelli F, Masè M. Computational mapping in atrial fibrillation: How the integration of signal-derived maps may guide the localization of critical sources. Europace 16 (5), pp. 714-723, 2014 (Review)
Ravelli F, Masè M, Cristoforetti A, Marini M, Disertori M. The logical operator map identifies novel candidate markers for critical sites in patients with atrial fibrillation. Progress in Biophysics and Molecular Biology 115 (2-3), pp. 186-197, 2014
Disertori M, Masè M, Marini M, Mazzola S, Cristoforetti A, Del Greco M, Kottkamp H, Arbustini E and Ravelli F. Electroanatomic mapping and late gadolinium enhancement MRI in a genetic model of arrhythmogenic atrial cardiomyopathy. Journal of Cardiovascular Electrophysiology 25 (9), pp. 964-970, 2014
Rieta JJ, Ravelli F, Sörnmo L. Guest Editorial: Advances in modeling and characterization of atrial arrhythmias. Biomedical Signal Processing and Control 8 (6), pp. 956-957, 2013
Disertori M, Quintarelli S, Grasso M, Pilotto A, Narula N, Favalli V, Canclini C, Diegoli M, Mazzola S, Marini M, Del Greco M, Bonmassari R, Masè M, Ravelli F, Specchia C, Arbustini E. Autosomal recessive atrial dilated cardiomyopathy with standstill evolution associated with mutation of Natriuretic Peptide Precursor A. Circulation: Cardiovascular Genetics 6 (1), pp. 27-36, 2013
Cristoforetti A, Mase M and Ravelli F. A fully adaptive multiresolution algorithm for atrial arrhythmia simulation on anatomically realistic unstructured meshes. IEEE Transactions on Biomedical Engineering 60 (9), art. no. 6514086, pp. 2585-2593, 2013
Ravelli F, Masè M, Cristoforetti A, Del Greco M, Centonze M, Marini M, Disertori M. Anatomic localization of rapid repetitive sources in persistent atrial fibrillation: Fusion of biatrial CT images with wave similarity/cycle length maps. JACC: Cardiovascular Imaging 5 (12), pp. 1211-1220, 2012
